The Leather Conservation Centre (LCC) in Northhampton, UK released some details of a new treatment for red rot. It reduces acidity and stabilizes the collagen molecules. Given the crushing number of red rotted bookbindings, and current lack of treatment options beyond surface consolidation, this is promising news. It will be interesting to keep an eye on this new product. At present, this treatment is only available at LCC and they are working out details of shipping, packaging, price, shelf life, and a name. The two products they currently sell for leather conservation are SC6000 and Selladerm.
…..
Investigation of Acid-Deterioration in Leather Leading towards Finding a Suitable Product for Treatment
Author/s (*indicate author for correspondence): Y Fletcher*1, A Lama1 , A P M Antunes2, and J Guthrie-Strachan2
Institutions:
1 The Leather Conservation Centre
2 The University of Northampton
Introduction
The purpose of the research was to carry out further investigation into acid-deterioration in leather in order to develop a product that can treat acid-deterioration with prolonged durability.
The two year research project into acid deterioration, with the aim of developing a new treatment, was carried out at The Leather Conservation Centre (the Centre) and the University of Northampton, both based in Northampton, UK. The University has an established leather technology degree programme. The project was a Knowledge Transfer Partnership (KTP) – a UK government backed scheme which links business and an academic institution enabling the business to access skills and expertise to help development of the business, in this case conservation of leather. Funded in part by the Centre and part by the UK’s Technology Strategy Board.
Acid-deterioration in leather, occurs in vegetable-tanned leathers that were predominantly manufactured from the mid-19 Century onward. Acid-deterioration has been observed in a variety of leathers such as bookbindings, gilt leather, screens, wall hangings, upholstery and luggage. The deteriorated leather shows a lower pH (≤3.0) and lower thermal stability. The visible changes usually include a powdery surface (often reddish/brownish, hence this particular deterioration is known as red-rot) and a complete or partial loss of the grain layer (the outer layer of leather); an acrid odour is also sometimes present.
However, care needs to be taken to ensure that a leather object does have acid deterioration and that the visible signs are not for other reasons.
For example, at the Centre we have been shown/given articles purporting to be suffering from “red rot” but which did not in fact have acid deterioration but had other deterioration mechanisms –
- heavy wear and tear – which had worn away the grain surface
- deliberate removal of grain surface – where much of the pigmented surface finish had cracked and flaked away, there had been a deliberate attempt to remove all the surface to give an even, though sueded, appearance to the leather surface
- delamination of the grain surface – a common deterioration mechanism in sheepskin
- suede and nubuck bookbindings – either a flesh split, or the surface had been deliberately abraded
- over application of leather dressings – causing blackening of surface with consequent flaking
It has long been accepted that both changes in the leather manufacturing processes and environmental pollutants (such as sulfur dioxide and nitrogen dioxide) are believed to be responsible for the acid-deterioration. There were considerable changes in leather manufacturing processes in the 19th Century to meet the increasing demand for leather; additionally, emission of sulfur due to the burning of coal gas at the time is also considered to be responsible for rapid deterioration.,
Conservation of acid-deteriorated historic leather has long been a concern due to the lack of suitable products for the treatment of acid-deterioration. The purpose of the research was to undertake further investigation into acid-deterioration in leather in order to develop a product that will delay the progress of the decay in acid-deteriorated historic leather.
For conservation purposes, it was decided that the ideal product should have collagen-stabilising properties, acid-buffering capacity, be capable of providing a long-term conservation effect to the acid-deteriorated leather, together with the fact that it would cause no damage to the leather or any other materials on a leather object with which it may come into contact. In addition any product should be safe for the conservators too (with correct use of any necessary PPE).
Materials and Methods
Desk-based research was conducted to review the products used in the past for the treatment of acid-deterioration. Based on the research and personal communication, various compounds were selected and trialled using new mimosa-tanned leather, artificially-aged mimosa-tanned leather and acid-deteriorated leather. Experiments were carried out using Mimosa-tanned leather (new and artificially-aged) and acid-deteriorated historic leather. Artificial ageing was carried out by exposing the leather samples to a concentration of sulfur dioxide (40-80ppm) and nitrogen dioxide (20-40ppm) at 40°C and 30% relative humidity for six and 12 weeks. Analyses were carried out at 2, 4 and 6 weeks for the six week trials, and at 3, 6, 9 and 12 weeks for the twelve week trials.
Differential Scanning Calorimetry (DSC) was used to determine shrinkage temperature (TS) which indicates changes in thermal stability of collagen and a pH test was used as an indication of acidity in the leather (new and artificially aged mimosa-tanned leather and acid-deteriorated historic leather) samples.
Reagents Trialled
The following chemicals were trialled –
- Aluminium diisopropoxide acetoacetate ester chelate (9.6% w/w Al) – the term aluminium alkoxide is used in this paper.
- 5-Ethyl-1-aza-3, 7-dioxabicyclo[3.3.0]octane – the term Organic Stabiliser is used in this paper.
A preliminary experiment was then carried out to determine the effects of the reagents on new mimosa-tanned leather. The products were then trialled on new mimosa-tanned leather that was previously artificially-aged for 6 weeks and on acid-deteriorated historic leather.
Shrinkage temperature (TS) and pH of the treated samples were determined. TS and pH of the corresponding untreated samples were also determined.
Solvents
White sprit is a non-polar organic solvent and so reduces the probability of solubilising polar components (salts and water soluble tannins) in leather when applied. White spirit is proven to be a safer solvent option and has been used in cleaning of historic leathers and therefore was chosen as a diluent for the purpose of this study.
Determination of shrinkage temperature (TS)
The leather samples were cut into small pieces (≈3 mg), placed in deionised water for a minimum of 16 hours at 20±2°C. The following day the excess of water was removed using Whatman No.1 filter papers and 5-10mg were placed in aluminium crucibles (40µl). The samples were analysed using Differential Scanning Calorimetry (DSC) (DSC822e, Mettler-Toledo, Switzerland). The initial and final temperatures during the analysis were 0°C and 100-150°C respectively at a ramping rate of 5°C/minute. The onset temperature of the denaturation process was recorded as TS.
Determination of pH
The pH of aqueous extract was determined following a British standard method (British Standard Institution; BS1309, 1974). A leather sample, 0.25±0.002g was placed in 5ml deionised water (pH: 6-7; adjusted using diluted sodium hydroxide) and agitated mechanically for 24 hours using a shaker at 20±2°C and 65±2% relative humidity. The following day the pH of the aqueous extract was measured using a standard pH meter.
Results and Discussion
Aluminium diisopropoxide acetoacetate ester chelate (aluminium alkoxide) was first introduced by Calnan (1989) for the treatment of acid-deterioration and investigated further during the STEPand ENVIRONMENT Leather Project, and was recommended as a treatment for acid-deterioration in leather. However, the effectiveness and longevity of this treatment were in doubt and the mechanisms of aluminium alkoxide against acid-deterioration are not entirely clear.
Earlier trials carried out at the Centre showed that aluminium alkoxide both increased the TS and pH of the new mimosa-tanned leather, artificially-aged mimosa-tanned leather and acid-deteriorated historic leather. The increase in TS and pH of new mimosa-tanned leather following the application of aluminium alkoxide indicates that the product has a collagen stabilising effect and an acid buffering capacity.
Research carried out at The British Museum showed that treatment of acid-deteriorated leather using aluminium alkoxide increased the pH from 2.7-3.0 to 4.6. However, it was reported that the pH of the treated leather decreased from 4.6 to 3.0-3.4 after 11 years. No information was found on the thermal stability of the experimented leather. As metal alkoxides are moisture sensitive, the replacement of an alkoxy group (O-R; R=alkyl group) through chelation is usually undertaken to reduce the hydrolysis rate.,, Aluminium alkoxide, although chelated, may hydrolyse completely over a period of time and therefore be unable, when applied on its own, to provide long-term protection against acid-decay.
The research showed that the aluminium alkoxide has an acid-buffering capacity and collagen stabilising property, as it increased the TS and pH of new mimosa-tanned leather, artificially-aged mimosa-tanned leather and acid-deteriorated historic leather. Retanning vegetable-tanned leather with aluminium is known to increase the thermal stability of vegetable-tanned leather. In this study, it appears that the Organic Stabiliser in the formulation reacts with alkoxide to enhance formation of stabilising matrix. The formulation may be creating a similar effect to retanning vegetable-tanned leather with aluminium.
A stabilisation effect on the artificially-aged mimosa-tanned leather and acid-decayed leather was obtained regardless of whether the Organic Stabiliser was applied before, after or in combination with aluminium alkoxide. However, increase in TS was found to be the highest when the Organic Stabiliser was applied in combination with aluminium alkoxide on artificially-aged mimosa-tanned leather.
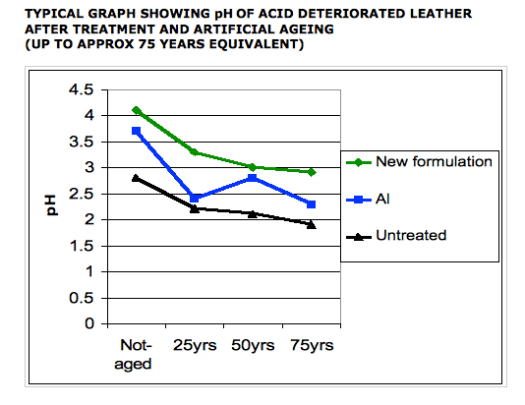
A formulation containing aluminium alkoxide and the Organic Stabiliser was found to be the optimal treatment for acid-deteriorated leathers. The study showed that the developed formulation not only buffers the acidity, but also increases the thermal stability. The stabilisation effect of the developed formulation on leather is higher than aluminium alkoxide when applied alone. Additionally, after artificial ageing in an acidic environment the artificially-aged mimosa-tanned leather and acid-deteriorated leather that were treated with the developed formulation exhibited a higher TS and pH than the respective untreated control samples (See Table 1).
The results of the trials, all carried out with the same method as given above showed that the new formulation reduced acidity (as evidenced by raised pH) and stabilised the collagen structure (as evidenced by raised TS) consistently.
During the study, it was observed that the higher the initial TS of leather, the higher the additive stabilisation effect conferred by the developed formulation. This suggests that treating acid-deteriorated leather at an early stage of deterioration could be beneficial.
It is expected that detailed results will be published in a peer-reviewed journal.
A change in appearance and firmness may occur due to the treatment. It was noted that on several samples the leather became darker and felt firmer/stiffer. Conservators will have to decide whether it is more important to retard the deterioration than to retain the colour and feel of the degraded leather. It should also be noted that acid-deteriorated leather is generally less firm and more flexible when compared to the new leather due to degradation, and the colour may also become lighter as a result of the decay. Therefore, it may be argued that the firmness and darkness observed after the application of the developed formulation may actually be bringing the leather closer to its original feel and appearance.
Humidification tests on a number of treated samples showed that the leather can be humidified after treatment to improve flexibility if required.
Any objects treated for acid deterioration with this new treatment will, almost certainly, need further remedial conservation treatment.
CONCLUSION
The benefits of the developed formulation are summarised as follows:
- Increase in pH
- Increase in thermal stability
- Capable of providing long-term protection against artificial ageing
- Once applied, further conservation treatments can be carried out.
- If necessary the leather can be consolidated (Cellugel was used in trials) before or after the Organic Stabiliser is applied.
Yvette Fletcher BA (Hons), MA, ACR
Head of Conservation
References
- Haines, B. M. (1980) The Structure, manufacture and Mechanisms of Deterioration of Book binding Leathers: Part 3, Minimising Deterioration in Polluted Atmospheres. In Conservation of Library and Archive Materials and the Graphic Arts: Abstract and Preprints, Cambridge 1980,. Institute of Paper Conservation and the Society of Archivists London, Guy Petherbridge.
- Larsen, R. (1995) Fundamental aspects of the Deterioration of Vegetable Tanned Leather. The Royal Danish Academy of Fine Arts School of Conservation.
- European Commission (1996) Environment Leather Project, Deterioration and Conservation of Vegetable tanned leather. Coordinator: R. Larsen. European Commission, Protection and Conservation of The European Culture Heritage, Research report no. 6. Denmark: L. P. Nielsen Offset Desktop Bogtryk.
- Larsen (2000) Experiments and Observations in the Study of Environmental Impact on Historical Vegetable Tanned Leathers. Thermochimica Acta, 365: 85-99.
- British Standard Institution (1974) Methods of Sampling and Chemical Testing of Leather, London, British Standard Institution.
- Calnan, C. N. (1989) Retannage with Aluminium Alkoxides-a stabilising Treatment for Acid Deteriorated Leather. The Leather Conservation Centre. Conference Proceeding, International Leather-and Parchmentsymposium International Committee of Museum (ICOM) Arbeitsgruppe, Leathercraft and Related Objects, 8 (12): 1989. Deutsches Ledermuseum, Frankfurt.
- European Commission (1994) STEP Leather Project, Evaluation of the Correlation between Natural and Artificial Ageing of Vegetable-Tanned Leather and Determination of Parameters for Standardization of an Artificial Ageing Method. R. Larsen (editor), Protection and Conservation of European Culture Heritage. Research report no. 1.
-
Lama, A. Antunes, A. P. M. Covington, A.D. Fletcher, Y. and Guthrie-Strachan, J.: A New Solution for the Treatment for Acid-Deteriorated Leather. Article submitted for publication.
- Parker, J. (2003) Re-evaluation of the condition of a previously conserved Mexican saddle and anquera. (Am.913), FR2003/22, Department of Conservation and Scientific Research, British Museum (unpublished),
- Haridas, M. M. & Bellare, J. R. (1999) Gellability zone for Aluminium Alkoxides. Ceramics International, 25 (7): 613-616.
- Arslan, O. Arpac, E & Sayılkan, H. (2010) Siliconcarbide Embedded Hybrid Nanocomposites as Abrasion Resistant Coating. Journal of Inorganic and Organometallic Polymers and Materials. 20 (2): 284-292.
- Lichtenberger, R. Puchberger, M. Baumann, S. O. & Schubert, U. (2009) Modification of Aluminum Alkoxides with β-Ketoesters: New Insights into Formation, Structure and Stability. Journal of Sol-Gel Science Technology, 50:130–140.
- Covington, A. D. (2009) Tanning Chemistry, The Science of Leather, Cambridge, RSC Publishing.